CSA Staff Notice 51-342 Staff Review of Issuers Entering Into Medical Marijuana Business Opportunities
CSA Staff Notice 51-342 Staff Review of Issuers Entering Into Medical Marijuana Business Opportunities
Introduction
Staff from the British Columbia Securities Commission, the Alberta Securities Commission, the Ontario Securities Commission and the Autorité des marchés financiers, (staff or we) recently reviewed the disclosure provided by certain reporting issuers that announced publicly their intention to enter into Canada's medical marijuana industry.
For the majority of issuers we reviewed, the disclosures in the original announcements were deficient, prompting staff to require the subsequent issuance of a clarifying disclosure document. This notice summarizes our overall findings and provides disclosure expectations for reporting issuers contemplating some involvement with the medical marijuana industry in Canada.
We also encourage issuers considering a change in business, not limited to medical marijuana opportunities, to use this notice as a tool to help ensure that any disclosure provided is factual and balanced.
Background
In June 2013, the Canadian Government enacted the Marihuana for Medical Purposes Regulations (MMPR) which govern the production, distribution and use of medical marijuana in Canada. These regulations became substantially effective on April 1, 2014.
There has been significant interest by the media, investors, and reporting issuers in this new area of business. In particular, we observed a substantial number of issuers announcing their intention to explore opportunities in the medical marijuana industry, following the introduction of MMPR.
A large majority of the issuers we reviewed in connection with this notice identified themselves as being in the junior mining industry prior to announcing their move to explore medical marijuana business opportunities. A breakdown of these issuers by their former identified industries is provided in the following chart:
In many cases, we observed that issuers who disclosed their intention to enter the medical marijuana industry obtained an immediate increase in their share price, even in cases where little, or any, substantive information was provided to the public about their prospective plans.
Given our concern that investors may face financial harm by purchasing an issuer's shares at an inflated price, before the issuer has established a viable business in the medical marijuana industry, we issued CSA Investor Alert: Caution Urged For Those Looking to Invest in Medical Marijuana Stocks on June 16, 2014.
Review Objectives
The objective of our review was to determine if issuers were meeting the requirements of National Instrument 51-102 Continuous Disclosure Obligations (NI 51-102) by providing sufficient and balanced disclosure related to their change in business to the medical marijuana field. In particular, we are concerned that issuers are benefiting from a positive impact on their share price by announcing their interest in the industry, but selectively omitting important information such as the stage of their plans and any risks and uncertainties related to those plans. Our review focused on whether an issuer's initial press releases, announcing its intent to enter this industry, contained sufficient detail to enable investors to understand what resources have been committed to the plan, as well as the related risks, cost implications and time required before the issuer can begin licensed operations. We are also concerned about whether press releases included unnecessary details such as exaggerated reports or promotional commentary, which could mislead investors as to the stage of development of an issuer's plans.
Review Scope
We initially reviewed the disclosure provided by 62 issuers announcing their intent to become involved in the medical marijuana industry. 40% or 25 of these issuers, raised serious investor protection concerns relating to balanced disclosure consistent with the issues outlined throughout this notice.
For the remaining 37 issuers, we were satisfied, for purposes of this review, that concerns around balanced disclosure and sufficient information to the public were mitigated through steps the issuers had taken announcing their change in business. For example, these issuers' business plans had progressed to a point where the issuer had been halted by their stock exchange in anticipation of a filing statement or other disclosure document in connection with an acquisition or a change of business. In these cases, more detailed and comprehensive information was to be provided in a disclosure document before trading would resume, which included a shareholder vote to approve the change in business transaction. As a result, these 37 issuers were determined to be outside the scope of this review. Once these issuers have completed their change of business transaction, they will be subject to our continuous disclosure review program.
The 25 issuers that we determined to be in scope for the purpose of this review were generally at a preliminary stage of entry into the medical marijuana field, including:
• Issuers in the early stages of due diligence that were contemplating medical marijuana opportunities in general terms, but had not committed to or disclosed any details about a specific opportunity or strategy for entering the industry;
• Issuers that filed a licence application with Health Canada under MMPR, or that acquired or invested in another company that applied for a licence; and
• Issuers that announced an agreement to invest in or acquire a medical marijuana business subject to certain terms, such as a non-binding letter of intent.
Regulatory Requirements and Guidance
NI 51-102 governs requirements on the timing and content of a reporting issuer's continuous disclosure record. Required disclosure items, such as management's discussion and analysis (MD&A) and material change reports, provide an opportunity for management of reporting issuers to discuss events which have had or may have a material impact on the issuer's performance. Part 1 (a) of Form 51-102F1 states, "Your objective when preparing the MD&A should be to improve your company's overall financial disclosure by giving a balanced discussion of your company's financial performance and financial condition including, without limitation, such considerations as liquidity and capital resources -- openly reporting bad news as well as good news".
National Policy 51-201 Disclosure Standards (NP 51-201) contains further guidance on the importance of providing balanced disclosure to investors in other disclosure documents, such as press releases. NP 51-201 states that "announcements of material changes should be factual and balanced. Unfavourable news must be disclosed just as promptly and completely as favourable news". The policy also states that "a company's press release should contain enough detail to enable the media and investors to understand the substance and importance of the change it is disclosing. Avoid including unnecessary details, exaggerated reports or promotional commentary".
When an issuer materially changes the focus of its business, it should ensure it communicates key information about its intended plans. This may include, but is not limited to, obtaining the appropriate licences or meeting regulatory requirements and determining whether the issuer has sufficient capital or other resources to implement the changes. The issuer needs to consider the level of disclosure to be included in a press release or material change report, which should include among other things, information about the time and resources required for the change in business as well as the barriers and obligations involved in realizing the change.
Issuers should also consider whether announcements about a potential change of business trigger the filing of a material change report as per the requirements of Part 7 of NI 51-102.
Findings and Outcomes
In general, we found that issuers' disclosure was often unbalanced and promotional in nature. While the benefits associated with involvement in the medical marijuana industry were often discussed, these discussions were not consistently accompanied by disclosure about the risks, uncertainties, cost implications and time required before the issuer can begin licensed operations.
Additionally, a discussion of barriers and obligations to entering the industry was often not provided. Given a majority of these issuers originated in industries other than medical marijuana, we are concerned that investors were not provided with sufficient information to understand the business changes being proposed by these issuers.
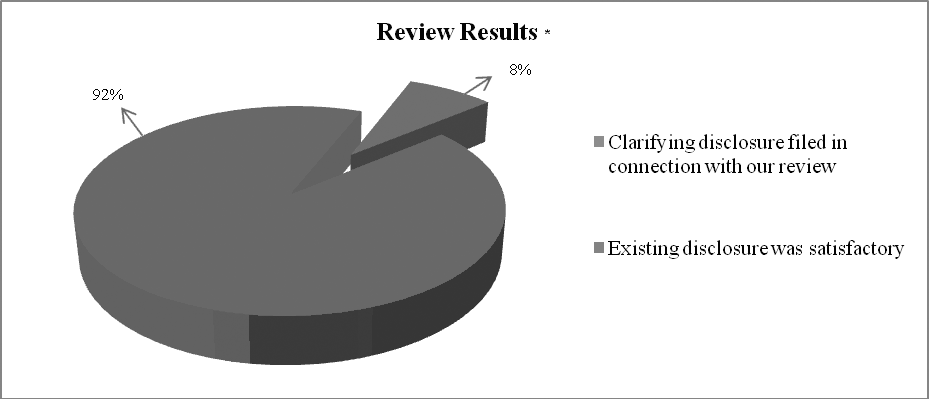
We sent comment letters to all issuers in the scope of our review. We asked 92% of these issuers to file a clarifying disclosure document in connection with our review, which they did. In most cases, issuers filed a clarifying press release which superseded the original announcements by providing more comprehensive and balanced disclosure. We were disappointed at the level of deficiency identified in the original announcements by issuers.
We identified the following specific disclosure deficiencies during the course of our review:
• Lack of a clear discussion of the issuer's stage of entry into the medical marijuana field, including development progress to date and steps that remain to be completed before a revenue generating medical marijuana business could begin (including the status of any licence application and whether or not an application had been submitted to date).
• No discussion of an estimate of the time, cost and nature of costs required to affect the proposed new business opportunity.
• Lack of any discussion of the medical marijuana licensing requirements of Health Canada, which are important in order for investors to adequately understand the resources and commitments which will be required in order to begin the new line of business.
• No discussion of any approvals (e.g., from the board of directors, shareholders or the issuer's securities exchange) which were obtained, or may still be required before the issuer may proceed with its proposed business plans.
• Failure to acknowledge that:
• The issuer will not be able to grow or sell medical marijuana without a licence from Health Canada.
• A facility meeting the licensing requirements of Health Canada must be available for inspection by Health Canada before a licence can be granted.
• There is no assurance that any prospective project in the medical marijuana industry will be successfully initiated or completed.
The following is an example of the deficient disclosure which we observed during our review, along with an example of how that disclosure could be enhanced.
- - - - - - - - - - - - - - - - - - - -
Example -- Deficient Disclosure
The Company's Board of Directors has approved a corporate diversification project whereby management has decided to enter the medical marijuana industry.
The Company has submitted its initial licence application to Health Canada and has entered into a non-binding letter of intent to purchase a facility in Sudbury, Ontario which will be used to produce medical marijuana.
The Company looks forward to participating in this new growth industry.
- - - - - - - - - - - - - - - - - - - -
- - - - - - - - - - - - - - - - - - - -
Example -- Enhanced Disclosure
Management is actively pursuing opportunities in the medical marijuana industry, including a medical marijuana licence under the Marihuana for Medical Purposes Regulations (MMPR).
As a condition of obtaining a licence, Health Canada requires significant steps to be taken, including the construction of an indoor growing facility equipped with physical barriers, visual monitoring, recording devices, intrusion detection, air filtration systems, as well as other important controls around distribution and access.
At this time, the Company has submitted a written licence application to Health Canada, however, no feedback has been received from Health Canada with respect to this application. As a result, none of the infrastructure required to support our licence application has as yet been ordered, purchased or assembled.
[If estimates of time and cost to complete the issuer's plans are not yet available]
Consequently, the Company is currently at too early a stage in its due diligence process to provide any estimate of the time or cost required in order to obtain a licence, or to assemble the infrastructure required in order to support our licence application. Until a facility meeting the requirements of MMPR is constructed, available for inspection by Health Canada and the Company is in receipt of a final licence from Health Canada, the Company cannot begin production of medical marijuana.
[If estimates of time or cost to complete the issuer's plans are available]
The amount of time required to obtain a licence is dependent on Health Canada's timeline for reviewing licence applications. Further, the amount of time the Company may need to resolve any comments received from Health Canada during the application process will not be known until such comments are received. As a result, the Company is currently at too early a stage in its due diligence process to provide any estimate of the amount of time required in order to obtain a licence. However, the Company has assembled a budget for the purchase of land in Sudbury, Ontario and the construction of a facility on that land which would meet the above noted licensing requirements of Health Canada. The Company currently anticipates costs of approximately $X in connection with these plans. The budgeted cost of the facility will be re-assessed once Health Canada has approved the design of the facility. Until a facility meeting the requirements of MMPR is constructed, available for inspection by Health Canada and the Company is in receipt of a final licence from Health Canada, the Company cannot begin production of medical marijuana.
Approval from the Company's Board of Directors, shareholders and stock exchange is also required before the Company may begin any operations in the medical marijuana industry.
There can be no assurance that the Company's medical marijuana licence application will be approved by Health Canada, or that any prospective projects in the industry will be successfully completed.
- - - - - - - - - - - - - - - - - - - -
If an issuer concludes that a previously announced initiative, such as the exploration of medical marijuana opportunities, is no longer being pursued then this information should also be disclosed.
Considerations for Other Industries
We remind issuers that the guidance in this notice is applicable to all industries, particularly companies thinking about material changes to their primary business or where an event has or will have an impact on future prospects. All issuers should ensure comprehensive, balanced disclosure is provided to investors, avoiding promotional commentary.
Conclusion
Announcements about significant events and business developments are often material information to investors. It is important to ensure that these announcements contain balanced disclosure about any associated risks, uncertainties or barriers to achieving the events being announced, rather than promotional commentary. Given their importance, we will continue to closely review these announcements, including as they relate to issuers exploring medical marijuana opportunities, through our continuous disclosure and prospectus review programs. We remind issuers that when we identify material disclosure deficiencies, we will request that the issuer correct the deficiency by filing clarifying disclosure. We may also consider further action depending on the circumstances.
Questions
Please refer your questions to any of the following:
Sonny Randhawa Jonathan Blackwell Manager, Corporate Finance Accountant, Corporate Finance Ontario Securities Commission Ontario Securities Commission 416-204-4959 416-593-8138 [email protected] [email protected] Oujala Motala Mike Moretto Accountant, Corporate Finance Manager, Corporate Disclosure Ontario Securities Commission British Columbia Securities Commission 416-263-3770 604-899-6767 [email protected] [email protected] Manny Albrino Cheryl McGillivray Senior Securities Analyst Manager, Corporate Finance British Columbia Securities Commission Alberta Securities Commission 604-899-6641 403-297-3307 [email protected] [email protected] Froshell Saure Martin Latulippe Securities Analyst, Corporate Finance Directeur, Direction de l'information continue Alberta Securities Commission Autorité des marchés financiers 403-355-3885 514-395-0337, ext. 4331 [email protected] [email protected] Pasquale Di Biasio Analyste, Direction de l'information continue Autorité des marchés financiers 514-395-0337, ext. 4385 [email protected]

